|
Developers:
|

|
Joseph Dinich
Beck School
Cherry Hill, New Jersey
|
Noel G. Harvey
Exploratory Plastics Research
Robin Queenan
Plastics Research
Rohm and Haas Company
Bristol, PA
|
|
|
|
Grade
Level:
|

|
7 through 9
|
|
|
|
Discipline:
|

|
Physical Science - Chemistry
|
|
|
|
Goal:
|
|
Students will identify density as a characteristic
property of matter.
|
|
|
|
Specific
Objectives:
|
|
Students will define density and
explain how to determine the density of an object.
|
|
|
|
Background:
|

|
Sometimes it is useful to know the
amount of mass in a given volume of an object.This quantity
is known as density. Density is defined as the mass per unit
volume of a substance. The following formula shows the
relationship between density, mass, and volume:
This demonstration shows the differences in the densities of
different liquids, and how these differences affect the
liquid's physical properties and interaction with other
materials.
|
|
|
|
Materials:
|
|
|
2 ice cubes
2 250-mL beakers
2 400-mL beakers
salt water solution
12-oz. diet soda
12-oz. regular soda
|
|
isopropyl alcohol
2 eggs
battery jar or aquarium
goggles
food coloring
|
|
|
|
|
Management
Tips:
|
|
- Add approximately 150 g of salt to 300 mL of water
for the salt solution.
- Use raw eggs. Also, use the same brand of diet and
regular soda.
- The density of water is l.00g/cm3 . Ice is 0.92g/cm3 ; sea water is 1.03g/cm 3 ;
alcohol is about 0.79 g/cm3.
- This activity can be adapted for use in cooperative
learning groups.
|
|
|
|
Procedure:
|

|
- This demonstration is a very
simple way to introduce and interest students in the
concept of density. Before the students arrive, prepare
the following examples of density differences as seen
through buoyancy:
a. Two 250-mL beakers for ice cubes - one with water, one
with isopropyl alcohol, or any nontoxic liquid with a
density of less than 0.92 g/cm 3 .
b. Two 400-mL beakers for eggs - one with 300 mL of
water, and one with 300-mL of a concentrated salt
solution.
c. An aquarium or battery jar of water for the soda cans
(save this for the following activity- �The Choice is
Yours: A Density Excursion�).
- Place the ice cubes, eggs, and
soda cans in the respective containers when you are ready
to perform the demonstration.
- After students make an initial
observation, add food coloring to each container to
improve visibility.
- Have students record and discuss
their observations and explanations leading to the
concept and definition of density.
- Explain how density is calculated
using sample problems.
a. If 96.6 g of gold has a volume of five cubic
centimeters, what is the density of gold?
b. If 96.5 g of aluminum has a volume of 35 cubic
centimeters, what is the density of aluminum?
- Have students make some mass and
volume measurements of the water and alcohol and
calculate their densities.
|
|
|
|
Questions:
|
|
- Explain why the eggs, ice cubes, and soda cans
floated in one liquid but not in the other.
- Why do you think oil floats on water?
- Give a stepwise procedure by which you could find the
density of any object.
|
|
|
|
References:
|
|
This activity was adapted from an experiment entitled
"The Mysterious Sunken Ice Cube" from the book Chemical
Demonstrations, Vol. 2, 2nd edition, Lee R. Summerlin,
Christie L. Borgford, and Julie B. Ealy, American Chemical
Society, 1988.
|
|
|
|
Extended Activities:
|
|
- The �Choice is Yours - a Density Excursion� may be
used as a follow up to this activity to further practice
calculating the density of matter.
- Determine the density of salt water.
- Using the substances and density information given,
show where the following substances would be located if
they were placed in a graduated cylinder by writing the
names of the objects on the lines provided in the
figure:
|
Air 0.001 g/cm3
corn oil 0.93g/cm3
corn syrup 1.38g/cm3
glycerine 1.26g/cm3
plastic 1.17g/cm3
rubber 1.34g/cm3
steel 7.81g/cm3
water 1.00g/cm3
wood 0.85g/cm 3
|
|

|
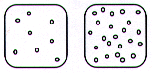
- Imagine that the squares below are beakers filled
with 100 mL of two different liquids, and that the dots
inside them are molecules of the different liquids. Draw
a circle around the square that contains the most dense
liquid.
|



